|
Similar to the pyrometallurgical method, the metals are recovered as inorganic salts. The hydrometallurgical route involves leaching disassembled or shredded LiBs in strong inorganic acids to dissolve metals and battery materials. After pretreatment, cobalt and manganese nickel are separated pyrometallurgically from the spent LiBs by melting and refining. The pyrometallurgical route is energy-intensive due to the high temperatures required. There are currently two basic industrially implemented technologies for the recycling of spent LiBs: the pyrometallurgical and the hydrometallurgical process 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11. Recycling is becoming increasingly important as many such batteries are at the end of their life cycle and the raw materials are expensive and are mined under critical conditions. Commercial cells are usually manufactured in a cylindrical design, whereas LiBs are used in the automotive industry in large prismatic cells or as so-called pouch cells.Īt the end of their lifetime, LiBs represent a valuable secondary raw material source to cover the demand of the necessary elements (Li, Ni, Mn, Co). Lithium-ion batteries (LiBs) are currently the most important technology for storing electrical energy and increasingly penetrating all areas of human everyday life due to their expanding use in smartphones, laptops, tools and e-mobility. Based on measurements of the temperature, voltage drop and copper concentration in the electrolyte at the cell with the originally lower charge capacity, the point of dissolution and incipient deposition of copper could be identified and a model of the processes during deep discharge could be developed. The current forced through the cell with a low charge capacity leads, after lithium depletion in the anode and the collapse of the solid-electrolyte-interphase (SEI) to a polarity reversal in which the copper collector of the anode is dissolved and copper is deposited on the cathode surface. If pouch cells with different states of charge are connected in series and deep-discharged together, copper deposition occurs preferably in the cell with the lower charge capacity. This paper is dedicated to the first step of each recycling process, the deep discharge of lithium-ion batteries, as a prerequisite for the safe opening and disassembling. Each process step in recycling must be performed in such a way contamination products on the cathode material are avoided or reduced. 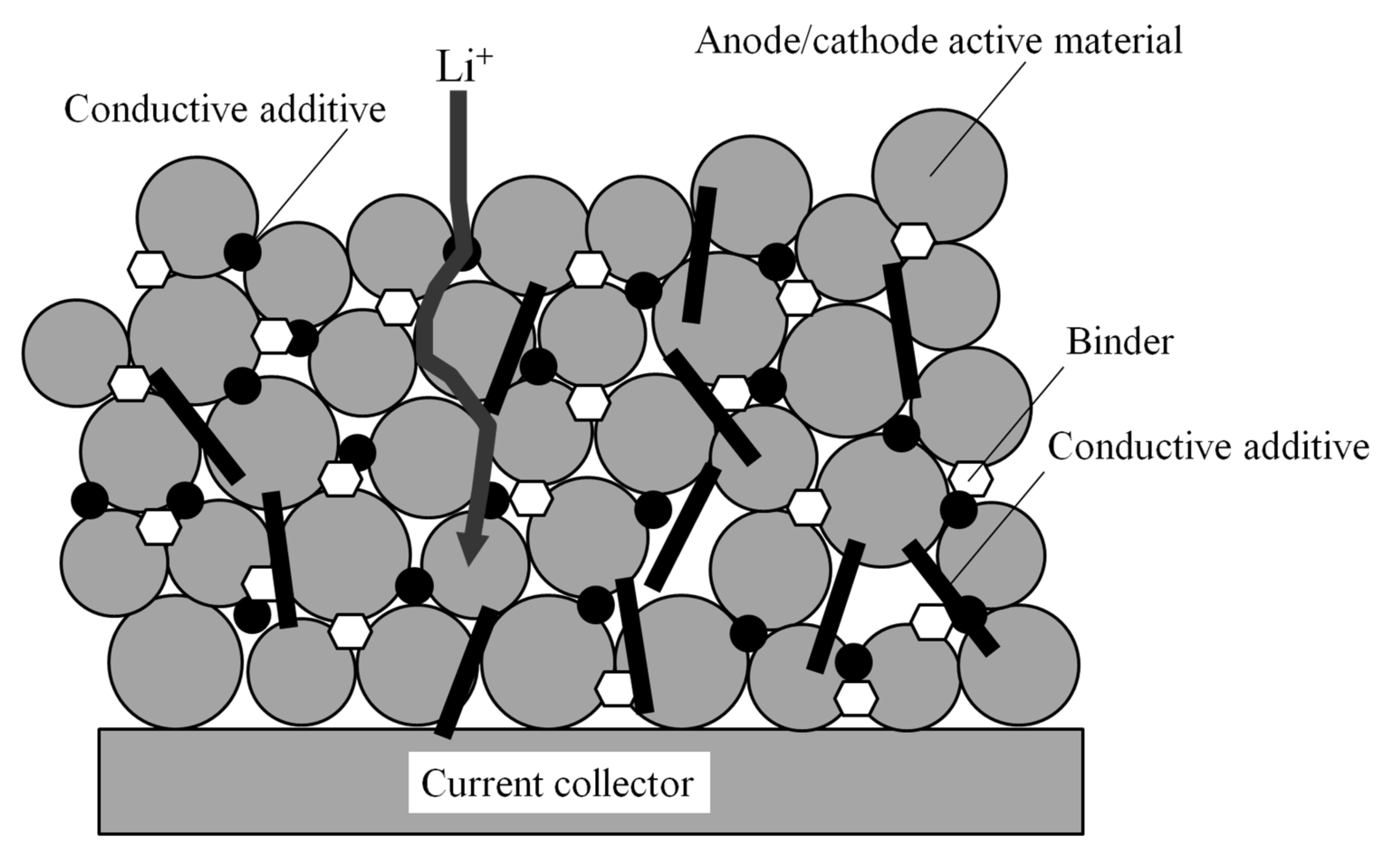 
End-of-life lithium-ion batteries represent an important secondary raw material source for nickel, cobalt, manganese and lithium compounds in order to obtain starting materials for the production of new cathode material.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
